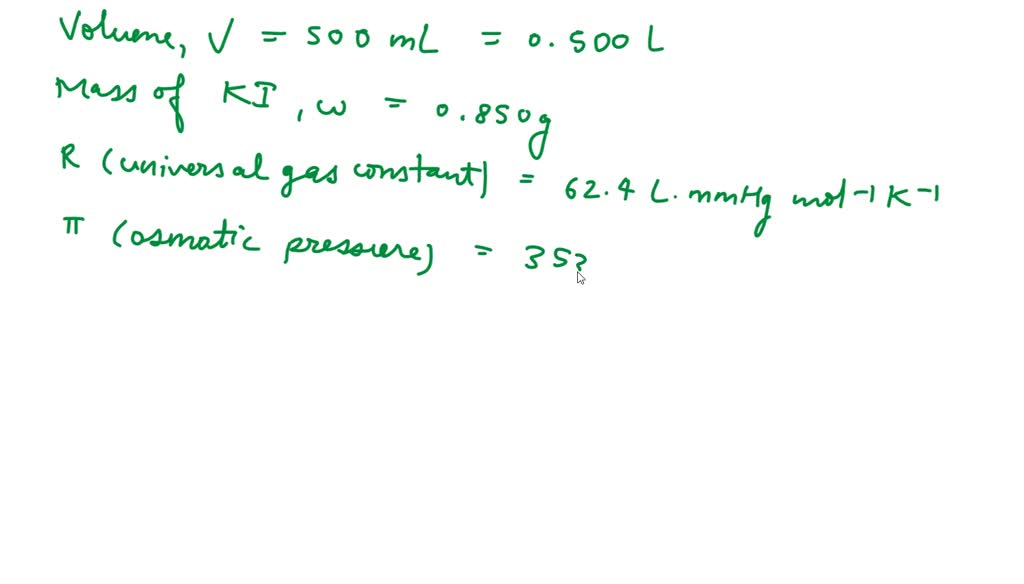Ki van't sales hoff factor
Finding the Van t Hoff factor sales, Solved Identify the solution with the highest van t Hoff Chegg sales, Van t Hoff Factor Definition Formula and Examples sales, Van t Hoff Factor sales, The osmotic pressure of 0.010 M potassium iodide solution KI at 25 sales, SOLVED Identify the solute with the lowest van t Hoff factor sales, Solved The experimental van t Hoff factor for KI at this Chegg sales, SOLVED Van t Hoff factor of a mixture of two moles of KI with 1 sales, The van t Hoff Factor Definition and How to Calculate It sales, SOLVED The osmotic pressure of a 0.0100 M potassium iodide KI sales, Solutions and Problems ppt download sales, Properties of Solutions. Solutions Solutions are homogeneous sales, Properties of Solutions. Solutions Solutions are homogeneous sales, Solution Van t Hoff factor sales, Solved Results Solute Molality Effective Molality ix m Chegg sales, 45. van t Hoff factor of a mixture of two moles of Kl with 1 mole sales, Chemistry Molality Van t Hoff Factor Applications Concept Video sales, The Van t Hoff factor of very dilute solution of Ca NO 3 2 sales, Solved 1. Which substance has the highest van t Hoff factor sales, SOLVED Identify the solute with the lowest van t Hoff factor. 0 sales, van t Hoff factor i 2 Now Tb i Kb m 2 0.52 1.0357 1.078 C sales, SOLVED Calculate the Van t Hoff factor for A 500.0 mL solution sales, Solved Determine the molarity of each of the following Chegg sales, The Van t Hoff Factor Unacademy sales, van t Hoff factor i 2 Now Tb i Kb m 2 0.52 1.0357 1.078 C sales, if p ka 4 then vant hoff factor for weak monobasic acid when c 0.01M sales, There is KI and sucrose solution with 0.1 M concentration if the osmotic pressure of KI and sucorse solution is 0.465 atm and 0.245 atm respectively. Then find the van t Hoff factor of KI and its sales, Van t Hoff plot showing the effect of temperature on the sales, The values of van t Hoff factors KCl NaCl and K 2SO 4 sales, In which case van t Hoff factor i remains unchanged a PtCl4 sales, What are the van t Hoff factors of C2H5OH no dissociation NaCl sales, SOLVED I will determine the molarity of each of the following sales, How To Calculate the VAN T HOFF FACTOR from the OSMOTIC PRESSURE sales, Calculate the dissociation constant of HCN if van t Hoft factor of sales, 03 93 ullysul always increases by doping If degree of ionisation sales, Equal volumes of 0.2M KI solution and 0.1M Hgl 2 solution are mixed sales, The osmotic pressures of 0.010 M solutions of KI and of sucrose C sales, Solved Choose the aqueous solution below with the lowest Chegg sales, SOLVED Calculate the Van t Hoff factor for A 500.0 mL solution sales, There is KI and sucrose solution with 0.1 M concentration if the osmo sales.
-
Next Day Delivery by DPD
Find out more
Order by 9pm (excludes Public holidays)
$11.99
-
Express Delivery - 48 Hours
Find out more
Order by 9pm (excludes Public holidays)
$9.99
-
Standard Delivery $6.99 Find out more
Delivered within 3 - 7 days (excludes Public holidays).
-
Store Delivery $6.99 Find out more
Delivered to your chosen store within 3-7 days
Spend over $400 (excluding delivery charge) to get a $20 voucher to spend in-store -
International Delivery Find out more
International Delivery is available for this product. The cost and delivery time depend on the country.
You can now return your online order in a few easy steps. Select your preferred tracked returns service. We have print at home, paperless and collection options available.
You have 28 days to return your order from the date it’s delivered. Exclusions apply.
View our full Returns and Exchanges information.